


Research Article
Grada van den Dool, Gonnie van Osta and Maarten Wiegerinck
Correspondence Address :
Maarten Wiegerinck
Wiegerinck Consultancy
Eindhoven, 5613CJ
The Netherlands
Received on: 29 March 2021, Accepted on: 12 April 2021, Published on: 14 April 2021
Citation: Grada van den Dool, Gonnie van Osta, Maarten Wiegerinck (2021) The FERTI•LILY Conception Cup Increases the Chance of Conceiving in Comparison with Expected Pregnancy Rates: A Prospective Observational Study
Copyright: 2021 Maarten Wiegerinck, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Background: With the average age of first-time mothers increasing over the last decades in the Western countries, difficulties to conceive naturally are rising in parallel.
The FERTI·LILY Conception Cup (FCC) is a medical device designed to stimulate natural conception. The aim of the current study was to evaluate the safety, efficacy, and user feasibility of the FCC.
Material and methods: 85 female volunteers trying to conceive entered the prospective, 1-arm, open label, non-randomized, observational study and were followed-up for maximally 3 months. The pregnancy rate observed in the study was compared to the pregnancy expectation based on 2 prognostic models to determine the device’s efficacy. Through questionnaires, information about safety, pregnancy, and user feasibility was collected.
Results: Of the 85 study participants, 65 used the product as instructed throughout the study. No Adverse Events (AEs) were reported. 23 participants conceived during the study, resulting in an overall pregnancy rate of 35.4% in the population that completed the study according to protocol, in contrast to the predicted rate of 23.9% (p < 0.05). Assessment of an infertile subpopulation resulted in a pregnancy rate of 30.3% in comparison with the predicted rate of 9.4% (p < 0.05). Study participants agreed that the FCC was comfortable and easy to use.
Conclusions: The FCC is a safe, comfortable, and easy to use medical device that significantly increases pregnancy rates, even in couples with fertility issues. A larger study is planned to provide additional data on pregnancy rates associated with the use of the FCC.
Keywords: Efficacy; FERTI·LILY Conception Cup; Medical device; Post-market evaluation; Pregnancy outcome; Safety
Introduction
The age of primigravida increased substantially over the last years, approaching 30 years in several European countries [1]. Although it is commonly believed that there are no substantial consequences from postponing pregnancy to a later age in life, it is clearly demonstrated that women’s day-specific probabilities of pregnancy declines with age starting from their late 20s [2-4]. With increasing age being the most powerful negative predictive factor of fertility, it is not surprising that between 8 to 12% of couples are confronted with infertility nowadays [5]. With infertility being defined as the failure to establish a clinical pregnancy after 12 months of regular and unprotected sexual intercourse. This observation is confirmed by a considerable increase in In Vitro Fertilization (IVF) in women over 35 years of age and the rise in average age of women attending the clinics for such purpose [6,7]. Also male fertility and sperm count in general is substantially reduced starting from 35 years of age and contributes for 20 to 30% to overall infertility rates [2,5,8].
Causes for infertility - if known – are diverse and include sperm abnormalities, ovulation dysfunction, and fallopian tube obstruction [9]. Usually, couples that have been trying to conceive unsuccessfully for 1 year or more are subsequently presented with a variety of treatment options ranging from hormonal treatments to regulate ovulation, to Intrauterine Insemination (IUI), Intra-Cytoplasmic Sperm Injection (ICSI), and IVF. Noteworthy, 50% of the couples with no known cause of infertility are still able to conceive naturally after a time of 12 months trying without successful pregnancy [10]. A possible explanation might be an inadequate transfer of the spermatozoa to the cervical mucus due to impairments of its functional competence, such as motility and velocity [11]. The proximity of the spermatozoa to the cervix is namely correlated to the number of spermatozoa reaching the cervical mucus, which increases the chances of conceiving [12]. One of the current standard techniques to maximize spermatozoa proximity to the site of fertilization is IUI, where sperm is directly transferred into the uterine cavity [13]. In addition, less invasive alternatives with such purpose have been explored, including cervical caps and the Stork® OTC Home Conception Device that physically increase the number of sperm cells near the cervical mucus and lead to considerable higher chances of conceiving [14,15]. However, these studies do not provide information regarding effects on the number of pregnancies.
Based on this mechanism of action a medical device, the FERTI·LILY Conception Cup (FCC, Rosesta Medical BV, The Netherlands) has been developed. The cup-shaped FCC is a CE marked Class I medical device consisting of 100% medical silicone and can be re-used up to 18 times or for 6 cycles. After natural intercourse, the device is physically self-inserted into the vagina promoting the ejaculate to reach the cervix. The device remains in place for 20 to 60 minutes to maximize spermatozoa reaching the cervical mucus. As such, the ejaculate remains in the
proximity of the cervix away from the potentially spermicidal vaginal environment after natural intercourse and as such improving the chances for a successful conception. The goal of the current study was to clinically evaluate the safety, efficacy, and ease of use of the FCC device in women that are trying to get pregnant.
Material and methods
Study design and population
The trial was designed as a prospective, 1-arm, open label, nonrandomized, observational study. All procedures performed in the current study are in accordance with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study population was derived from women that spontaneously purchased the FCC device. Inclusion criteria to enter the study included female sex, willingness and ability to give written consent for study participation, no ongoing pregnancy at the time of entering the study, intention to use the FCC, and trying to conceive by intercourse with a male partner. Study participants were informed about the study in a neutral manner using non-technical language about the purpose of data collection. Participation was voluntarily and informed consent with regard to participation and use of data was obtained from all study participants prior to enrolment.
Sample size analysis was based on a literature search focusing on fertility treatments in a general population [16,17]. The expected pregnancy was based on the retrieved data and calculated to be 22% after 3 months of unprotected intercourse. Sample size analysis estimated an n-value of 60 to be sufficient to estimate the proportion of participants becoming pregnant and to preclude events not occurring in the study with sufficient precision for comparison with literature (two-sided test; alphalevel of 0.05; power = 80%). Considered an average drop-out rate of 10% during the study, and a 30% drop-out before study initiation and preliminary study exit due to pregnancy, a total number of 100 participants was calculated.
The study was carried out by an independent company, DJEM Insight Studio (Enkhuizen, The Netherlands), specialized in online research. At time of study initiation, each participant received a test package for the duration of 1 menstrual cycle using the FCC, consisting of a non-digital ovulation and a non-digital pregnancy test (Swiss Precision Diagnostics GmbH) to track their fertile days and determine positive or negative pregnancy outcome, respectively. The use of the ovulation and pregnancy test was voluntary. 1 month after inclusion the subjects were requested to fill in an anonymous questionnaire relating to safety, pregnancy outcome, and usability of the FCC. Questions related to user feasibility of the medical device involved subject’s satisfaction, ease of use, comfort, and size of the FCC. Volunteers that did not respond to this questionnaire were excluded from the study. For subjects not pregnant after the first month but with the intention to continue using the FCC, test material was provided for another 2 months of ovulation and pregnancy testing. At the end of the study, which was 3 months after inclusion or at the time point of a positive pregnancy test, participants were requested to fill in the questionnaire again.
Study outcomes
Participants’ demographics
At the start of the study, relevant data was collected including demographics, fertility-related medical history, number of months trying to conceive (unprotected intercourse), and prior pregnancies.
Safety evaluation
Before study initiation, an Ethical Evaluation was carried out. Since the FCC CE-marked medical device is used within the intended purpose for which CE marking was issued (BMH, article 13, and the BAI, article 2), the current study falls within exception to the notification obligation. All required quality, safety, and efficacy issues for the test medical device have been addressed and evaluated based on the documentation essential to fulfil the requirements of the European Council Directive 93/42 EEC. In addition, according to the Risk Management Procedure for the medical device in compliance with ISO 14971, no unacceptable risks have been identified with an overall acceptable residual risk level.
The batch and serial number of the supplied FCC devices were recorded for each participant to correlate with potential Adverse Events (AEs). Any AEs related or not related to the medical device under investigation were reported within 3 days from the acknowledgment to the Sponsor.
Efficacy
Efficacy of the FCC was assessed by evaluation of the number of pregnancies at the end of the study. This pregnancy rate was compared with 2 prognostic models that predict pregnancy outcome both in the general population (Sozou et al.) and in a subpopulation of infertile couples trying to conceive for at least 1 year (Hunault et al.) [18,19]. As such, included subjects served as their own control where their predicted pregnancy rate was compared to the observed pregnancy rate.
The Sozou model provides a pregnancy prediction in the general population by calculating the chance of conceiving based on the intrinsic conception rate (ICR). The ICR is defined as a couple’s probability of achieving a pregnancy in the next cycle. The ICR takes female age and the number of months trying to conceive into account. As such, study participants were appointed to age groups based on the subdivision of Sozou et al., where 4 specific ages were assessed including 25, 30, 35, and 40 years of age. In the current study, participants were appointed to age categories, resulting in three age groups: 30 (age between 21 and 29 years old), 35 (age between 30 and 34 years old), and 40 (age over 35 years old). The number of months trying to conceive were rounded up to the nearest period (0, 1, 3, 6, 9, 12, 24, or 36 months) based
on the definition of Sozou et al. The following formula was used to determine the probability of conceiving for each study participant:
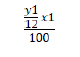
Where x1 is the number of assessed cycles and y1 is the ICR as determined by the Sozou model. The number 12 represents the minimal number of menstrual cycles per year which varies between 12 and 13.
Both in literature and daily practice, a differentiation is made between couples trying to conceive for less than 1 year and couples who fail to establish a clinical pregnancy after this period despite regular unprotected intercourse. For this specific subpopulation, Hunault et al. developed a prognostic model which was validated by van der Steeg et al. through a largescale prospective study [20]. The pregnancy prediction rate for a subgroup of study participants that were trying to conceive for 12 months or more was evaluated using the following formula:
 Where x2 is the number of assessed cycles, y2 is the probability score for each couple as defined by Hunault, and 12 the yearly menstrual cycles. Calculations of the probability score were performed through an on-line calculator based on the Hunault model (https://www.freya.nl/probability.php). The score takes female age, duration of non-conception, and previous pregnancies into account. In addition, it was assumed that no post-coital test was performed, there was no tubal pathology, total time of trying to conceive was capped at 12 months, referral was done by a general practitioner, and sperm motility was set at 55%. The reported average sperm motility in couples trying to conceive for over 12 months is 35% [21].
Where x2 is the number of assessed cycles, y2 is the probability score for each couple as defined by Hunault, and 12 the yearly menstrual cycles. Calculations of the probability score were performed through an on-line calculator based on the Hunault model (https://www.freya.nl/probability.php). The score takes female age, duration of non-conception, and previous pregnancies into account. In addition, it was assumed that no post-coital test was performed, there was no tubal pathology, total time of trying to conceive was capped at 12 months, referral was done by a general practitioner, and sperm motility was set at 55%. The reported average sperm motility in couples trying to conceive for over 12 months is 35% [21].
Statistical analysis
The proportion of pregnant women was calculated using the following formula: (100*Number of women becoming pregnant)/Number of women in the population. Exact 95% confidence intervals (Clopper-Pearson) of the proportion of women becoming pregnant for the various (sub)populations in the study population were calculated.
Statistical analysis was performed using SAS® for WindowsTM version 9.4.and data were presented as mean ± SD or as percentage. Descriptive statistics were calculated for demographic characteristics.
Results
Study population
The study was conducted between November 2019 and July 2020, with an individual subject duration between one and three months depending on the speed of achieving pregnancy and length of the menstrual cycle. 1384 women that purchased the FCC were approached by email querying their interest in the study participation. Of the 182 respondents, 123 women fulfilled the predetermined inclusion criteria of which 85 were willing to participate in the study. Of these 85 participants, 10 women indicated a positive pregnancy test 1 month after study initiation. 13 of the non-pregnant volunteers did not provide further information about potential pregnancies or the use of the FCC for the rest of the study duration. At the end of menstrual cycle two, 5 women were pregnant and 57 were not pregnant, of which 7 discontinued the study. At the end of the study after cycle three, 8 women reported to be pregnant and 42 were not. In total, 65 study participants completed the study protocol as planned and 20 did not complete or return the questionnaire after cycle 1 or 2. Women that did not know whether they were pregnant at the time of completing the questionnaires, were considered not pregnant. An overview of the investigational process is shown in Figure 1.
A differentiation was made between the population that completed the study according to the protocol by using the FCC for 3 cycles or until pregnancy (n=65, ‘protocol population’) and the intention-to-treat population (n=85, ‘total population’, which also included volunteers that only completed 1 or 2 cycles of the study. In addition, a subdivision for each group was made for couples trying to conceive for at least 1 year in both the ‘total population’ and ‘protocol population’ group, represented by the ‘infertile total‘ (n=43) and ‘infertile protocol’ (n=33) population (Figure 2).
Study participants had an average age of 30.3 ± 3.5 years, ranging from 21 to 41, evenly distributed over the total and infertile populations. 21.2% of the women indicated in the questionnaire to have fertility problems, such as low sperm motility and sperm count, and unregular menstrual cycles because of polycystic ovary syndrome. This percentage was considerably higher in the infertile subpopulations where 30.2% of the total population and 27.3% of the protocol population were confronted with fertility issues. Three-quarters of the participants were nulliparous. Subject demographics are summarized in Table 1.
Safety
Prior to study initiation, usability studies have shown that the FCC can be inserted, worn, and removed as intended without any unforeseen risks. The likelihood of a clinical benefit to the patient hereby outweighs the risks associated with use of the device. Throughout the study, there was only 1 AE reported, ectopic pregnancy, which was unrelated to the study device.
Efficacy
The general pregnancy expectancy for the current total and protocol study population was calculated with the Sozou model of which details can be found in Supplemental table 1. The average pregnancy outcome expectation was 18.1 pregnancies and 15.5 pregnancies for the total study population and the protocol population, respectively, resulting in calculated pregnancy probabilities of 21.3% and 23.9% over three months.
In the current study, a total of 23 pregnancies was recorded leading to a pregnancy rate of 27.1% in the total population and a significantly higher pregnancy rate in the protocol population of 35.4% (p < 0.05).
When looking at the chance of conceiving per menstrual cycle, 23 out of 197 evaluated cycles resulted in a pregnancy and subsequently a pregnancy probability of 11.7% per cycle in the total population. Similarly, evaluation of the 23 pregnancies over 170 cycles in the protocol group resulted in a pregnancy rate of 13.5% per cycle.
Hunault’s model estimates the chance of pregnancy after trying to conceive for 12 months or more over a period of 3 menstrual cycles to be 8.3% for the total infertile population, which is equivalent to 3.5 expected pregnancies. As for the infertile protocol subpopulation a pregnancy chance of 9.4%, or 3.1 expected pregnancies, was calculated. Calculation details can be found in Supplemental table 1. In the current study, 10 pregnancies were reported in the infertile subpopulation, leading to a subgroup average pregnancy rate of 23.3% and 30.3% in the total and protocol infertile population, respectively. The observed pregnancy rates in the current study significantly outnumbers this prediction both in the total infertile and infertile protocol population (p < 0.05).
When looking at the number of cycles evaluated resulting in the 10 observed pregnancies, a pregnancy probability per cycle of 6.9% was calculated for the total infertile population (104 cycles) and 11.1% for the infertile protocol population (90 cycles). Predicted and observed pregnancies are summarized in Figure 3 over the total study duration
Although the observed pregnancy rate in the infertile group was slightly lower than in the total study population, the difference was not statistically significant. In addition, there was no significant correlation observed between age or previous pregnancies of the study populations with the pregnancy rate in the general and infertile study populations. This might be due to the limited number of study participants.
User feasibility
Overall, the FCC comfort and its use were experienced positive by the study participants. As such, 85% of the total study population reported no difficulty in using the product, 98% of users experienced no discomfort while using the product, and 82% of users found the size of the cup (just) right. Only 2% of users thought the cup was ‘a little too big’. Results of the user feasibility are visualized in Fig. 4.
Discussion
The age of primigravida has been increasing over the last years, which is known to be one of the factors contributing to infertility. More and more couples are having difficulties to conceive, which leads to stress, anxiety, marital problems, and even physiological issues, considerably impacting their quality of life [22]. Various reproductive treatment options are available to date, but these may induce more stress, can be costly, often require assistance from a physician, or might disturb the natural conceiving process, while no success is guaranteed [23].
The mechanism of action of the FCC under evaluation is based on promoting natural conception through physical guidance of the ejaculate near the cervix after intercourse. Thus increasing the number of sperm cells near the cervical mucus and promoting the chance to conceive [14,15]. The current study demonstrates that 35.4% of the women using the medical device during every menstrual cycle were pregnant after 3 months, which is significantly more than the 23.9% pregnancy prediction rate in this population.
Results obtained for a subpopulation that has been trying to conceive for at least 1 year were even more pronounced. In this infertile population the calculated pregnancy probability of 8.3% in the total study population and 9.4% in the population that completed the study according to protocol, was statistically significant outweighed by the observed pregnancy rates of 23.3% and 30.3%, respectively. In addition, use of the FCC is even progressing toward pregnancy rates induced by IUI as demonstrated by a retrospective study where this technique results in 39% clinical pregnancies in couples with unexplained infertility [13]. Overall, the use of the FCC was indicated as easy and comfortable while no device-related AEs were reported throughout the study.
In conclusion, the current exploratory post-market clinical evaluation shows how use of the FCC increases the chance of conceiving both in a general population and in a population with decreased fertility. The FCC might thus provide an inexpensive, low threshold, and effective alternative for couples who are trying to conceive without the need for specialized, invasive reproduction therapies. Future assessments of a larger study group shall provide more data on pregnancy probability rates and more insight in potential correlations with age, fertility problems, and previous pregnancies in the general population and specific subgroups.
Funding
Rosesta Medical BV sponsored this study and is also the manufacturer of the investigational product. The study was independently designed and performed by DJEM Insight Studio and funded by Rosesta Medical BV.
Compliance with ethical standards
All procedures performed in the current study are in accordance with the 964 Helsinki declaration and its lateramendments or comparable ethical standards. Informed consent for study participation and use of the collected data was obtained from all study participants prior to their inclusion in the study.
Disclosure of interest
MW is medical advisor and shareholder to Rosesta Medical BV and collaborated in the development of the medical device under investigation, supported the design of the current clinical evaluation, and served as the principal investigator of the study.
Acknowledgements
The authors would like to thank all the volunteers that participated in this study and Jan Willem van der Steeg for the careful review of the prognostic model as comparator for the pregnancy rates in the infertile subpopulation used in the study. Writing of the manuscript was performed by an independent medical writer, Leni Vandekerckhove (MediGraphX, info@ medigraphx.be).
1. Eijkemans MJC, Van Poppel F, Habbema DF, et al. Too old to have children? Lessons from natural fertility populations. Hum Reprod. 2014;29:1304-1312.
2. Dunson DB, Colombo B, Baird DD. Changes with age in the level and duration of fertility in the menstrual cycle. Hum Reprod. 2012;17:1399-403.
3. Keefe D, Kumar M, Kalmbach K. Oocyte competency is the key to embryo potential. Fertil Steril; 2015; p. 317–22.
4. Hart RJ. Physiological aspects of female fertility: Role of the environment, modern lifestyle, and genetics. Physiol Rev. 2016;96:873-909.
5. Vander Borght M, Wyns C. Fertility and infertility: Definition and epidemiology [Internet]. Clin. Biochem. 2018;p. 2-10.
6. Bellieni CV. Neonatal risks from in vitro fertilization and delayed motherhood. World J Clin Pediatr. 2012;1:34.
7. Andersen AN, Goossens V, Ferraretti AP, et al. Assisted reproductive technology in Europe, 2004: results generated from European registers by ESHRE. Hum Reprod. 2008;23:756–71.
8. Levine H, Jørgensen N, Martino-Andrade A, et al. Temporal trends in sperm count: A systematic review and meta-regression analysis. Hum Reprod Update. 2017;23:646–59.
9. Koroma L, Stewart L. Infertility: Evaluation and Initial Management [Internet]. J Midwifery Women’s Heal. 2012;p. 614–21.
10. Gelbaya TA, Potdar N, Jeve YB, Nardo LG. Definition and epidemiology of unexplained infertility. Obstet Gynecol Surv. 2014;69:109-15.
11. Schats R, Aitken RJ, Templeton AA, Djahanbakhch O. The role of cervical mucus-semen interaction in infertility of unknown aetiology. BJOG An Int J Obstet Gynaecol. 1984;91:371-376.
12. Whitelaw WJ. The cervical cap self-applied in the treatment of severe oligospermia. Fertil Steril. 1979;31:86–7.
13. Michau A, El Hachem H, Galey J, et al. Predictive factors for pregnancy after controlled ovarian stimulation and intrauterine insemination: A retrospective analysis of 4146 cycles. J Gynecol Obstet Hum Reprod. 2019;48:811–5.
14. Alexander NJ. Evaluation of male infertility with an in vitro cervical mucus penetration test. Fertil Steril. 1981;36:201-208.
15. Pelekanos MJ. Postcoital Sperm Assessment Comparative Study. Surg Technol Int. 2015;27:184–90.
16. Evans-Hoeker E, Pritchard DA, Long DL, et al. Cervical mucus monitoring prevalence and associated fecundability in women trying to conceive. Fertil Steril. 2013;100.
17. Tiplady S, Jones G, Campbell M, Johnson S, Ledger W. Home ovulation tests and stress in women trying to conceive: A randomized controlled trial. Hum Reprod. 2013;28:138–51.
18. Sozou PD, Hartshorne GM. Time to Pregnancy: A Computational Method for Using the Duration of Non-Conception for Predicting Conception. PLoS One. 2012;7.
19. Hunault CC, Habbema JDF, Eijkemans MJC, et al. Two new prediction rules for spontaneous pregnancy leading to live birth among subfertile couple, based on the synthesis of three previous models. Hum Reprod. 2004;19:2019-26.
20. van der Steeg JW, Steures P, Eijkemans MJC, et al. Pregnancy is predictable: A large-scale prospective external validation of the prediction of spontaneous pregnancy in subfertile couples. Hum Reprod. 2007;22:536–42.
21. Cooper TG, Noonan E, von Eckardstein S, et al. World Health Organization reference values for human semen characteristics. Hum Reprod Update. 2009;16:231–45.
22. Chachamovich JR, Chachamovich E, Ezer H, et al. Investigating quality of life and health-related quality of life in infertility: a systematic review. Journal of Psychosomatic Obstetrics & Gynecology. 2010; 31:2.
23. Suarez SS, Pacey AA. Sperm transport in the female reproductive tract. Human Reproduction Update. 2006; p. 23–37.
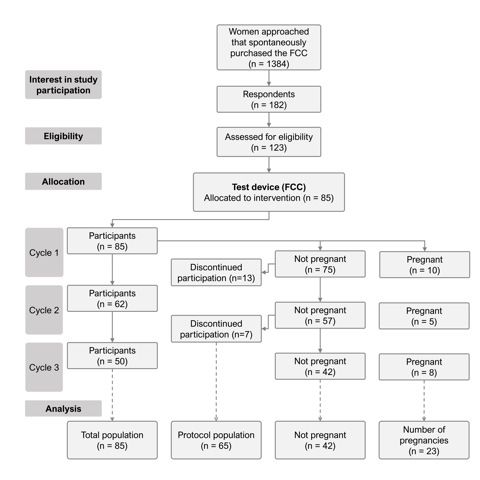
Figure 1: Flow diagram providing an overview of the investigational process. The ‘total population’ represents all study participants that completed at least 1 cycle according to protocol. The ‘protocol population’ includes all study participants that completed the study according to protocol and used the investigational device for 3 cycles or until clinical pregnancy was established.
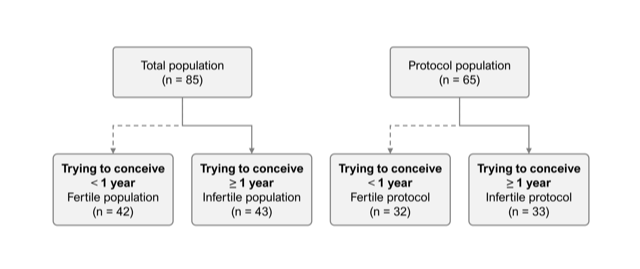
Figure 2: Flow diagram presenting the infertile subpopulation in the total and protocol populations.
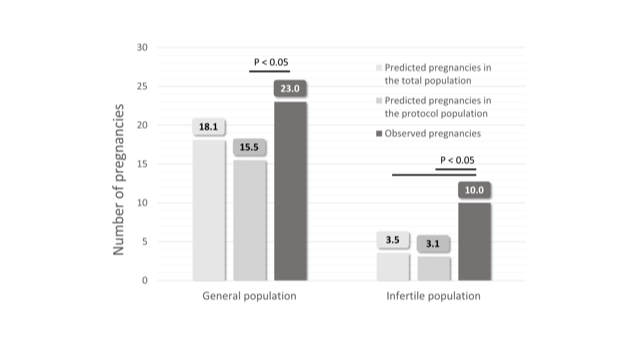
Figure 3: Overview of the predicted and observed pregnancies over the total study duration in the general and infertile study populations.
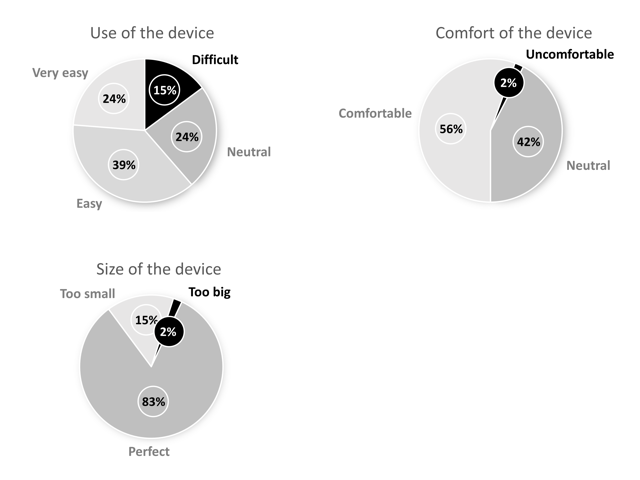
Figure 4: Summary of the user feasibility results, including ease of use, comfort, and size of the medical device.
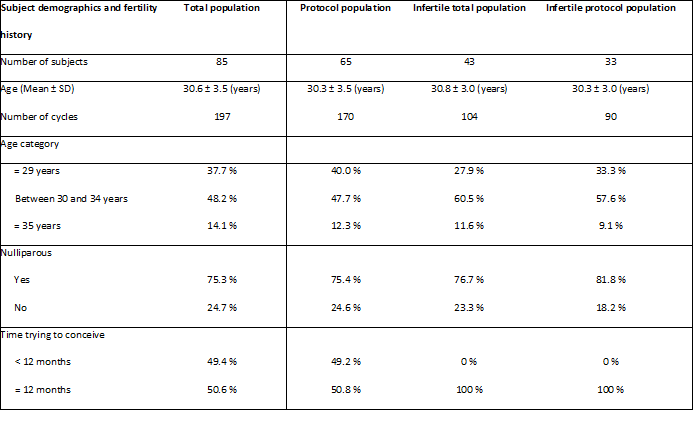
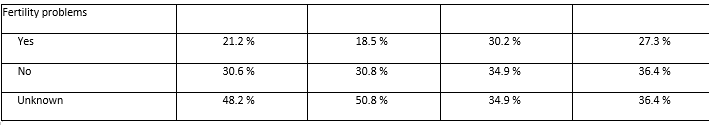
Table 1: Subject demographics and fertility history
 Open Access
by Scient Online is licensed.
Open Access
by Scient Online is licensed.